This three part post on etching is in conjunction with my upcoming demo for the Metal Clay Artisan Guild in Connecticut.
The above etched cuff is a great way to start learning to etch. It was a scrap of copper roofing material that was "sandwiched" in masking tape. I cut the design out with an exacto knife from the tape and used an aquatinting technique for the resist in the background to create the texture. If you follow the link here to my last blog post, you can learn all about different resist techniques to get your design on the metal.
As I discussed in my previous post, my etching experiences began a very long time ago with materials like nitric acid and asphaltum. Today there are much safer etching and resist solutions. For etching in this post, I am using Ferric Chloride, also known as the "salt" etch. Ferric Chloride is not an acid, so rather than dissolving the metal, it agitates it and pieces fall out. Here is some more information from Printmaking Today that discusses the use of Ferric Chloride in the art studio, just follow this link.
Supplies
I use the following materials when etching:
-Saefty Glasses, Rubber Gloves & an Apron
-2 Plastic/Disposable Bowls
-Floral Foam or a Sponge (a disposable object that floats)
-Duct Tape
-Ammonia
-Your prepared Copper
-Ferric Chloride (also known as "PCB Etchant")
Ferric Chloride can be purchased online from a number of jewelry supply companies, but if you can find it locally at a Radio Shack, you will avoid a hazardous shipping fee. Also, I prefer to purchase chemicals in the smallest quantities necessary until I know how much I am going to use. This saves me from have a huge stock of a chemical that I need to responsibly dispose of. Radio Shack sells it for etching motherboards and a pint is around $11. For jewelry work, a pint will last you quite a while. Follow this link here to check it out at Radio Shack, if it's not in stock you should be able to select "ship to store" for free.
Safety First!
Ferric Chloride is a salt and not an acid, so you do not have to worry about it being absorbed by the skin. It also does not give off or produce toxic fumes, so is a great alternative. With that being said, the rule of thumb for all chemicals is: Safety Glasses, Apron, Rubber Gloves, Close Toed Shoes and Ventilation! On a side note, it will stain everything it comes into contact with a dark, sludge green color. It will take weeks to get it off your skin and nails and it will not come out of clothes or countertops, which is why despite its safety you really want to wear gloves and an apron. As you pour it and work with it, consider the same precautions you would take with a large bowl of liquid black ink. The black ink will stain less.
Prepare to Etch

The first time you etch you really need to do a test strip to see how fast your etchant is working and to determine how deep you want the etch to be. For the discs with the birds on it in this series of posts, I used 22 gauge copper and wanted to find an etch depth that would be deep enough to use as a texture plate in metal clay. I cut a test strip out of copper the same thickness as the final pieces I was doing. Coat the back with clear nail polish to protect it. On the front, using a permanent marker as a resist, draw a border and divide the piece into 4 boxes. Number each box (1-2-3-4) with the marker as a resist. You can see my finished test on the right, it has a textured background. I did 30 minute increments, took the piece out, rinsed it then covered each number/segment with duct tape to protect it from further etching and placed the piece back in the solution. If I have not etched in a while or I am etching a new project, I always run test strips. It is a cheap way to ensure I do not ruin a piece, it only takes a few extra minutes to etch all the way through and have a hole in your piece!

 To etch the piece, pour the Ferric Chloride solution into a plastic or glass container, 1" should be more than deep enough. Once an item is used in the studio, it can never go back to the kitchen, so these containers should be marked or disposed of, this is an easy clean up so they can be reused for more etching in the future. Keep it small - the container only needs to be slightly larger than what you are etching. Cut a piece of floral foam or a sponge a little bigger than your piece. Wrap a piece of duct tape around the foam with the sticky side OUT. Wrap another piece around three of the four sides to cover the sticky sides so you can handle it. Use rubber gloves to place your piece with its back pressed firmly into the sticky tape. Remember, at this point the oils from your fingers could ruin your design. Then place the piece in the container and give it a press. It should bounce back and gently float around. Since the salt agitates, but does not dissolve the metal, it is a good idea to tap it or gently agitate it while it is etching to make sure the pieces are falling out and giving a clean etch. While your piece is etching, get a bucket of water to rinse it in and have it partly filled next to your solution.
To etch the piece, pour the Ferric Chloride solution into a plastic or glass container, 1" should be more than deep enough. Once an item is used in the studio, it can never go back to the kitchen, so these containers should be marked or disposed of, this is an easy clean up so they can be reused for more etching in the future. Keep it small - the container only needs to be slightly larger than what you are etching. Cut a piece of floral foam or a sponge a little bigger than your piece. Wrap a piece of duct tape around the foam with the sticky side OUT. Wrap another piece around three of the four sides to cover the sticky sides so you can handle it. Use rubber gloves to place your piece with its back pressed firmly into the sticky tape. Remember, at this point the oils from your fingers could ruin your design. Then place the piece in the container and give it a press. It should bounce back and gently float around. Since the salt agitates, but does not dissolve the metal, it is a good idea to tap it or gently agitate it while it is etching to make sure the pieces are falling out and giving a clean etch. While your piece is etching, get a bucket of water to rinse it in and have it partly filled next to your solution.Final Etch
 When the time is up, based on your test strips, pull the piece out and rinse it in the water bucket. Next take a clean sponge with some Ammonia on it and rub the piece to stop the etch from continuing. Pull the pieces off the foam. The pieces may need to be clean with acetone (or nail polish remover) to get the rest of the resist off. You can see here that the etched piece has a "starburst" effect in the background. Since the metal is not being dissolved, large areas are often left with this effect. The best way to control a large open area is with the aquatint resist technique. I find that Ferric Chloride solution has about 5 hours of etching time for me, which equates to 2-3 times of use. I also use it all at the same time, so I might etch some pieces, cover the solution and then be sure to either use it or dispose of it within a week. After that, you just don't know if you will get the same results from it and I don't think it's worth the risk of ruining a piece with potentially spent etchant. I have successfully used ferric chloride from the same original bottle for years, so it does have a long shelf life until activated. Check out the final blog post in this series to see how these copper discs can be used as texture plates.
When the time is up, based on your test strips, pull the piece out and rinse it in the water bucket. Next take a clean sponge with some Ammonia on it and rub the piece to stop the etch from continuing. Pull the pieces off the foam. The pieces may need to be clean with acetone (or nail polish remover) to get the rest of the resist off. You can see here that the etched piece has a "starburst" effect in the background. Since the metal is not being dissolved, large areas are often left with this effect. The best way to control a large open area is with the aquatint resist technique. I find that Ferric Chloride solution has about 5 hours of etching time for me, which equates to 2-3 times of use. I also use it all at the same time, so I might etch some pieces, cover the solution and then be sure to either use it or dispose of it within a week. After that, you just don't know if you will get the same results from it and I don't think it's worth the risk of ruining a piece with potentially spent etchant. I have successfully used ferric chloride from the same original bottle for years, so it does have a long shelf life until activated. Check out the final blog post in this series to see how these copper discs can be used as texture plates.Disposal
The Ferric Chloride should be neutralized with baking powder. Allow any solids to settle and drain off any neutralized liquid into your rinse bucket so that it is diluted and flush down the toilet. The remaining solids or sludge should be poured into a plastic sealable container, clearly labeled, and disposed of at your local hazardous waste disposal facility.



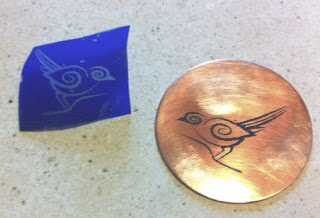
No comments:
Post a Comment